 Attended polysomnography, which allows sleep stage, arousal, and body position determination, is the “gold standard” of PAP titration. The technologist can also address patient complaints, mask or mouth leaks, hypoxemia, or significant arrhythmias. Education about OSA and PAP, mask fitting, and a period of PAP acclimatization should be performed before lights out. Some of the common problems encountered during PAP titration and their solutions are listed in Table 2.
Attended polysomnography, which allows sleep stage, arousal, and body position determination, is the “gold standard” of PAP titration. The technologist can also address patient complaints, mask or mouth leaks, hypoxemia, or significant arrhythmias. Education about OSA and PAP, mask fitting, and a period of PAP acclimatization should be performed before lights out. Some of the common problems encountered during PAP titration and their solutions are listed in Table 2.
PAP titration requires proper interpretation of information. The flow signal from the analog or digital output of the PAP device derived from an accurate internal flow sensor can be used to detect reductions in flow or flattening (airflow limitation). A flattened airflow profile suggests residual upper airway narrowing. A leak signal output is also available for recording and helps identify unintentional leak (mask or mouth). Widely fluctuating leak without a pressure change or body movement suggests presence of an oral leak, which can mimic hypopneic events (Fig 5), prevent airway stabilization, or result in arousal from sleep. The optimal end point of titration has not been scientifically determined. Most laboratories titrate to eliminate obstructive apnea, hypopnea, snoring, oxygen desaturation, and respiratory effort-related arousals. The target AHI is usually < 5 to 10/h, although some centers also try to eliminate airflow limitation even if not associated with arousal. As noted above, the highest pressures are usually needed in the supine posture during REM sleep. Therefore an ideal PAP titration is one that demonstrates control of events in the supine position during REM sleep.
One CPAP titration approach is to increase pressure in 2 cm H2O increments for apnea or 1 cm H2O for hypopnea/snoring at pressures 12 cm H2O. A starting pressure of at least 4 cm H2O is required to generate sufficient leak via the antirebreathing aperture to prevent carbon dioxide buildup. Some patients may find it difficult to initiate sleep if the initial pressure is too low. Excessively rapid upward titration does not allow the patient to adjust to positive pressure and leaves the physician who reviews the titration wondering if a lower pressure might have been effective. For bilevel PAP titration, an approach would be to start with 8/4 cm H2O and then increase both IPAP and EPAP as needed to abolish obstructive events. In general, EPAP is increased to abolish obstructive apneas and the IPAP-EPAP difference increased to abolish hypopneas, snoring, and oxygen desaturation. It is difficult to stabilize the upper airway of patients with OSA using too low an EPAP or too high an IPAP-EPAP differential. Of note, a different strategy is used for noninvasive ventilation of patients without significant OSA. In this case, a relatively low EPAP (5 to 6 cm H2O) and high IPAP-EPAP difference (10 to 12 cm H2O) is often successful.
Complex OSA is a term that has been used to identify OSA patients in whom central apneas persist or develop during PAP titration. Treatment-emergent central apnea is another term for central apneas that appear during CPAP titration. This type of central apnea occurs during NREM sleep because the PaC02 falls below the apneic threshold. A central apnea may follow an increase in ventilation due to a preceding arousal or an underlying instability in ventilatory control. Central apneas were in fact well described after treatment of OSA with tracheosto-my and tended to resolve with time. Such resolution of central apneas can also occur in some patients with long-term CPAP treatment. Thomas and co-workers described a group of patients with obstructive apnea who displayed a cyclic alternating pattern in the EEG and persistent events during NREM sleep that were not eliminated by increases in pressure. As the pressure level was increased, a “break point” was noted with the emergence of repetitive central apneas. Of interest, these patients did not improve with chronic PAP treatment. The best treatment approach for treatment-emergent central apnea/complex sleep apnea is currently unknown. One can try a reduction in pressure in a patient who previously had fair control of obstructive events (without central apneas) at lower pressures. A slight increase in pressure could be tried if respiratory effort-related arousals are triggering subsequent central apneas. Sometimes, sleep-onset central events will resolve during the study once stable sleep is reached. While technologists often switch from CPAP to bilevel PAP in an effort to abolish central apnea, this can actually worsen the situation unless a backup rate is used. There have been preliminary reports of success in patients with complex sleep apnea with ASV.
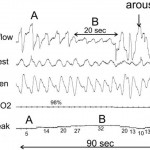
Figure 5. A 90-s tracing during a PAP titration. A period of increased leak (B) was associated with a drop in the CPAP flow signal. Note that the flow profile remains rounded in B unlike what is usually seen during obstructive hypopneas. With arousal (C), the leak suddenly dropped. There was minimal body movement from A to C (mask did not move). Video monitoring revealed a mouth opening during B (reprinted with permission from Berry RB, Sleep Medicine Pearls, second edition, Philadelphia, PA: Hanley and Belfus, 2003; 130).
Table 2—Problems During CPAP Titration and Typical Solutions
| Problems | Solutions |
| Claustrophobia | Desensitization; nasal pillows; nasal prong interface |
| Mask leak | Careful mask fitting before study; adjust straps; change mask size or type |
| Nasal congestion | Full-face mask; heated humidity; decongestant |
| Pressure intolerance | Avoid rapid increase in pressure; temporary reduction in pressure to allow return to sleep; bilevel PAP; flexible PAP; elevation of head of bed, side sleep position to decrease required pressure |
Category: Apnea
Tags: autoadjusting positive airway pressure, continuous positive airway pressure adherence, obstructive sleep apnea, positive airway pressure treatment